







The Verneuil process, also called flame fusion, was the first commercially successful method of manufacturing synthetic gemstones, developed in 1902 by the French chemist Auguste Verneuil.
It is a process for making synthetic rubies, sapphires, spinels, etc., by the fusion at high temperatures of powdered compounds. Also called flame-fusion process.
The Verneuil process or slight modifications of the process can be used to create a wide variety of materials, corundum, spinel, strontium titanate, etc. The description below is based on it's first use to create ruby and sapphire.
The idea of "flame grown" corundum dates back to about 1886-1888. This is the time that Auguste Victor Louis Verneuil began his pioneering work with melting aluminum oxide in a hydrogen-oxygen flame.
His work came to fruition in about 1904 when he finally perfected the process. Today, the process has only been changed slightly from the original process with mainly better temperature control attributing to the more effective growth of the boules.
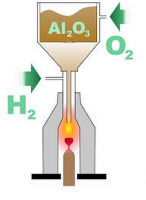
The process begins with a seed crystal of corundum placed on a movable platform below a burner head, and in a well insulated cylindrical kiln.
The burner consists of concentric tubes. The center tube is used to provide the alumina (aluminum oxide) growth powder and the colorant. In the case of ruby the red color is caused by chromium oxide. Sapphire colorant (dark blue) is created from a mixture of iron oxide and titanium dioxide.
The exterior tube (highlighted in yellow here) provides the oxygen and hydrogen gas to make the flame. The process must be run at over 2050 C to be successful.
Here the feed powder is started and directed toward the seed crystal.
The flame is now ignited and the process begins. The alumina in the gas phase is only partially melted. The majority of the growth takes place by the melting of the alumina on the surface of the seed crystal.
It is necessary to maintain strongly oxidizing conditions throughout the growth. Hence it is not unusual to use extra oxygen in the process to keep everything bathed in an oxidizing atmosphere.
As the burning process continues the stage with the seed crystal, and now growing boule, is slowly lowered and pull downward away from the burner. The rate of withdrawl is about 1 cm per hour.
The flame is turned off at the end of the growth cycle and the boule either shatters in the next 10 or so seconds or survives. The process produces a good deal of internal stress. If the boule survives, it is taped on top and the last of the internal stress is relieved as it breaks almost cleanly in half, producing "half boules".