







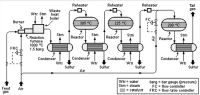
The Claus process is a catalytic chemical process for converting gaseous hydrogen sulfide (H2S) into elemental sulfur (S).The process is commonly referred to as a sulfur recovery unit (SRU) and is very widely used to produce sulfur from the hydrogen sulfide found in raw natural gas and from the by-product sour gases containing hydrogen sulfide derived from refining petroleum crude oil and other industrial facilities.
There are many hundreds of Claus sulfur recovery units in operation worldwide. In fact, the vast majority of the 66,000,000 metric tons of sulfur produced worldwide in 2006 was by-product sulfur from petroleum refining and natural gas processing plants.
Early History
In nineteenth century, there were many alkali manufacturing plants in England producing sodium carbonate (Na2CO3) by the Leblanc process. The original Claus process was developed by Carl Friedrich Claus, a chemist working in England, for the purpose of recovering sulfur from the waste calcium sulfide (CaS) generated by the Leblanc process. As a catalyst, he chose a bog iron ore and later bauxite (a mineral with a high alumina content).In 1883, Claus was granted a British patent for the process.
During the next 53 years, the Claus process underwent several minor modifications. In 1936, I.G. Farbenindustrie a (German conglomerate of chemical companies) introduced a modification of the process that utilized a thermal conversion step followed by catalytic conversion steps, which is the basically the concept currently used in modern Claus sulfur recovery units.